UofSC chemists look to the future of materials science
Published on May 15, 2020
Laura Murdock ’16 likes the thrill of creative chemistry ― making new materials that could, in turn, make a difference in the world.
After finishing her undergraduate degree in chemistry at South Carolina, she came back to earn a PhD and do research in synthetic chemistry. “I felt like it was something impactful,” she says. “You can make something that would look very small on paper, but actually makes a big difference.”

One of her latest creations is like that. It’s a thin, yellow-brown film. A small piece that she stretches between her gloved hands in the lab looks like old plastic wrap. Actually, it’s the world’s best known material for filtering carbon dioxide and methane from each other. And the method she used to make it could help materials scientists fight climate change or reduce pollution.
The new membrane resulted from teamwork by Murdock; her advisor, Brian Benicewicz, the University of South Carolina SmartState Chair in Polymer Nanocomposite Research; and scientists at Columbia University in New York. They wanted to develop a better way to design and make membranes that separate gases. Those materials could be used to filter greenhouse gases and pollutants, but they are generally designed and tested with a trial-and-error approach.
Their new method, published today in Science Advances, uses big data to improve the process.
It removes the guesswork and the old trial-and-error work, which is very ineffective. You don't have to make hundreds of different materials and test them.
― Brian Benicewicz, SmartState professor
Tradeoffs
Benicewicz explained that gas-filtering membranes suffer from a tradeoff between selectivity and permeability ― a material that lets one gas through is unlikely to stop a molecule of another gas. “We're talking about some really small molecules,” Benicewicz said. “The size difference is almost imperceptible. If you want a lot of permeability, you're not going to get a lot of selectivity."
The team at Columbia University created a machine learning algorithm that analyzed the chemical structure and effectiveness of existing membranes used for separating carbon dioxide from methane. Once the algorithm could accurately predict the effectiveness of a given membrane, they turned the question around: What chemical structure would make the ideal gas separation membrane?
Sanat K. Kumar, the Bykhovsky Professor of Chemical Engineering at Columbia, compared it to Netflix’s method for recommending movies. Like Netflix identifies features a viewer enjoys in movies and then recommends movies that share that feature, his team’s algorithm found which chemical structures contributed to effective membranes.
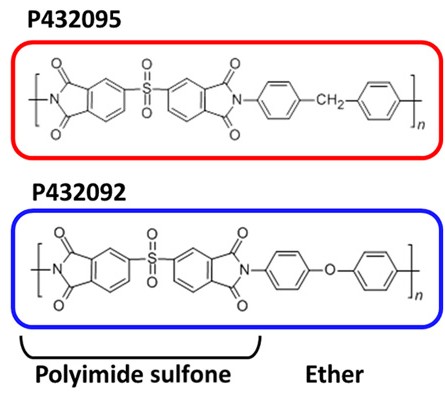
The computer produced a list of 100 hypothetical materials that might surpass current limits. Benicewicz, who leads a synthetic chemistry research group, identified two of the proposed structures that could plausibly be made. Murdock’s job was to make them.
You can make something that would look very small on paper, but actually makes a big difference.
― Laura Murdock, PhD candidate
Outstanding results
Making the materials involved mixing substances called monomers with a solvent. They reacted and formed polymers with a repeating chemical structure. Murdock fashioned the polymers into thin membranes.
Murdock admits to being excited and nervous when she sent the materials to be tested. Would they separate CO2 and methane as well as she hoped?

The test results could hardly be better. The membranes’ effectiveness was close to the computer’s prediction and well above presumed limits.
“Their performance was very good ― much better than what had been previously made," Murdock said. “And it was pretty easy. It has the potential for commercial use.”
Separating carbon dioxide and methane has an immediate application in the natural gas industry; CO2 must be removed from natural gas to prevent corrosion in pipelines. But Murdock said the method of using big data to remove the guesswork from the process leads to another question: “What other polymer materials can we apply machine learning to and create better materials for all kinds of applications?”
Benicewicz said machine learning could help scientists design new membranes for separating greenhouse gases from coal emissions, which could help to reduce climate change.
“It removes the guesswork and the old trial-and-error work, which is very ineffective,” Benicewicz said. “You don't have to make hundreds of different materials and test them. Now you're letting the machine learn. It can narrow your search.
About the Study
The study is titled: “Designing exceptional gas-separation polymer membranes using machine learning.”
The authors are: J. Wesley Barnett, Connor R. Bilchak, Yiwen Wang and Sanat K. Kumar from the Columbia University Department of Chemical Engineering; Brian C. Benicewicz and Laura A. Murdock from the University of South Carolina Department of Chemistry and Biochemistry; and Tristan Bereau from the Max Planck Institute for Polymer Research.
The study was supported by the National Science Foundation Graduate Research Fellowship Program (grant DGE-16-44869 to C.R.B.), the Designing Materials to Revolutionize and Engineer our Future Program (grant 1629502 to C.R.B., J.W.B., and S.K.K. and grant 1629052 to B.C.B. and L.A.M.), and the Emmy Noether Program of the Deutsche Forschungsgemeinschaft (to T.B.). S.K.K., J.W.B., and C.R.B. acknowledge that the majority of the work was funded by NSF through DMREF grant CTS-1629502. This project was started under a previous NSF grant (DMR150730), which was continued as grant DMR-1929655.